Update on screening for Barrett's and esophageal cancer

This review nicely updates the status of screening programs for Barrett's esophagus and esophageal adenocarcinoma, with and emphasis on established and newer non-invasive methods.
The Horizon of Screening for Barrett's Esophagus and Esophageal Cancer
Techniques and Innovations in Gastrointestinal Endoscopy,
2023 https://doi.org/10.1016/j.tige.2023.01.004.
https://www.sciencedirect.com/science/article/pii/S2590030723000120
Tan WK, Rebecca C. Fitzgerald RC.
Abstract
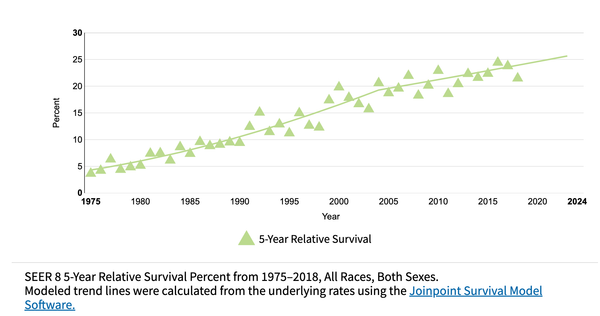
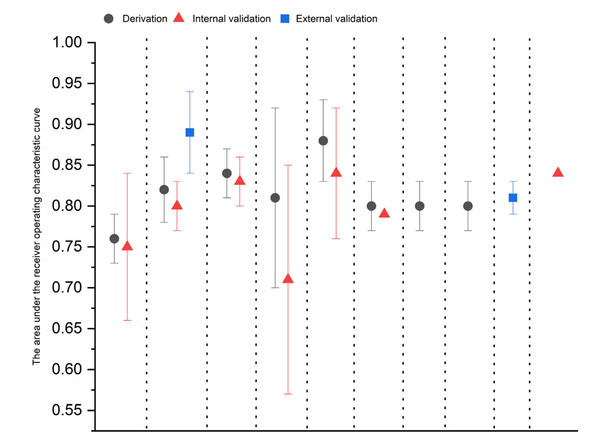
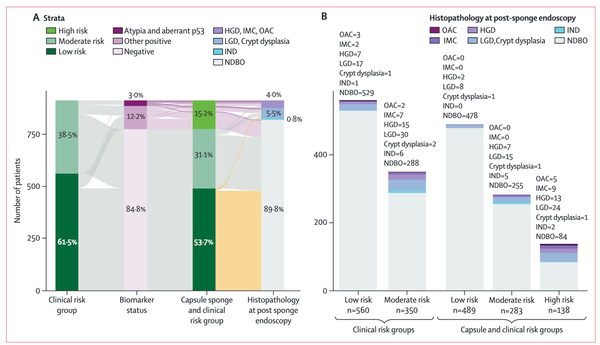
Barrett's esophagus (BE) is a pre-malignant precursor to esophageal adenocarcinoma (EAC), a cancer whose incidence has increased 6-fold in the western world over the past three decades. The prognosis associated with EAC remains poor, with the 5-year survival estimated to be <20%. Epidemiological and clinical study evidence has suggested that early detection and treatment of BE-related neoplasia is associated with improved survival, suggesting that screening for this condition could impact EAC survival. Over the past few decades, there have been accelerated advances in the field of BE and EAC, particularly in the emergence of non-endoscopic cell-collection devices that could be used for screening. In this review, we critically discuss the concept of screening BE and EAC as well as identifying the target population who should be screened. We then review the evidence for various cell-collection devices which could be used for screening in the office-based setting, such as the Cytosponge and biomarker Trefoil-factor 3 (TFF3), EsophaCap and methylated DNA markers (MDMs) as well as the balloon-based EsoCheckTM and MDMs. We also discuss other novel technologies such as volatile organic compounds (VOC) detection using the electric-nose, and technologies utilizing optical coherence tomography. These promising technologies have paved the way for the potential introduction of a screening program for BE and EAC, with the hope that this could lead to improved outcomes among patients who suffer from this unfortunate disease.
Keywords: Barrett's esophagus; Esophageal adenocarcinoma; screening; non-endoscopic devices