Update on Barrett's screening
While not breaking new ground, this manuscript from Mayo Clinic nicely reviews state of the art approaches to screening (and surveillance) for Barrett's esophagus and esophageal adenocarcinoma. These newer techniques including: three different swallowable cell sampling devices combined with biomarkers (now approved in society guidelines), capsule-based imaging, and analysis of molecular biomarkers from peripheral blood samples (e.g., circulating tumor DNA, circulating mRNA.)
Advances in Screening for Barrett Esophagus and Esophageal Adenocarcinoma.
Vantanasiri K, Kamboj AK, Kisiel JB, Iyer PG.
Abstract
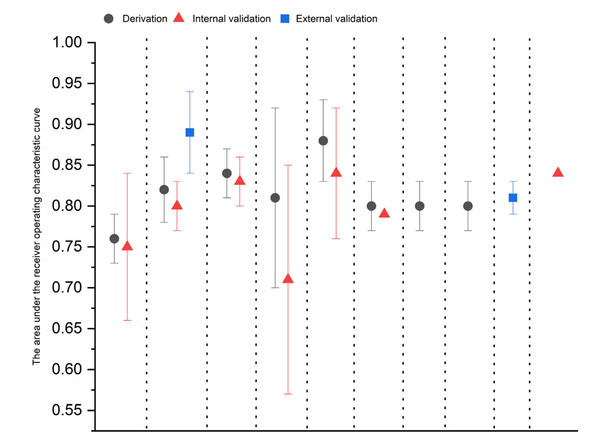
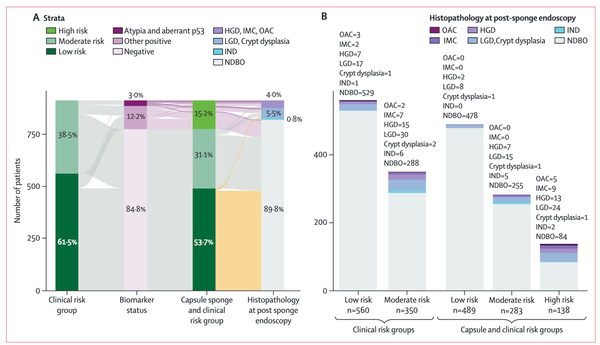
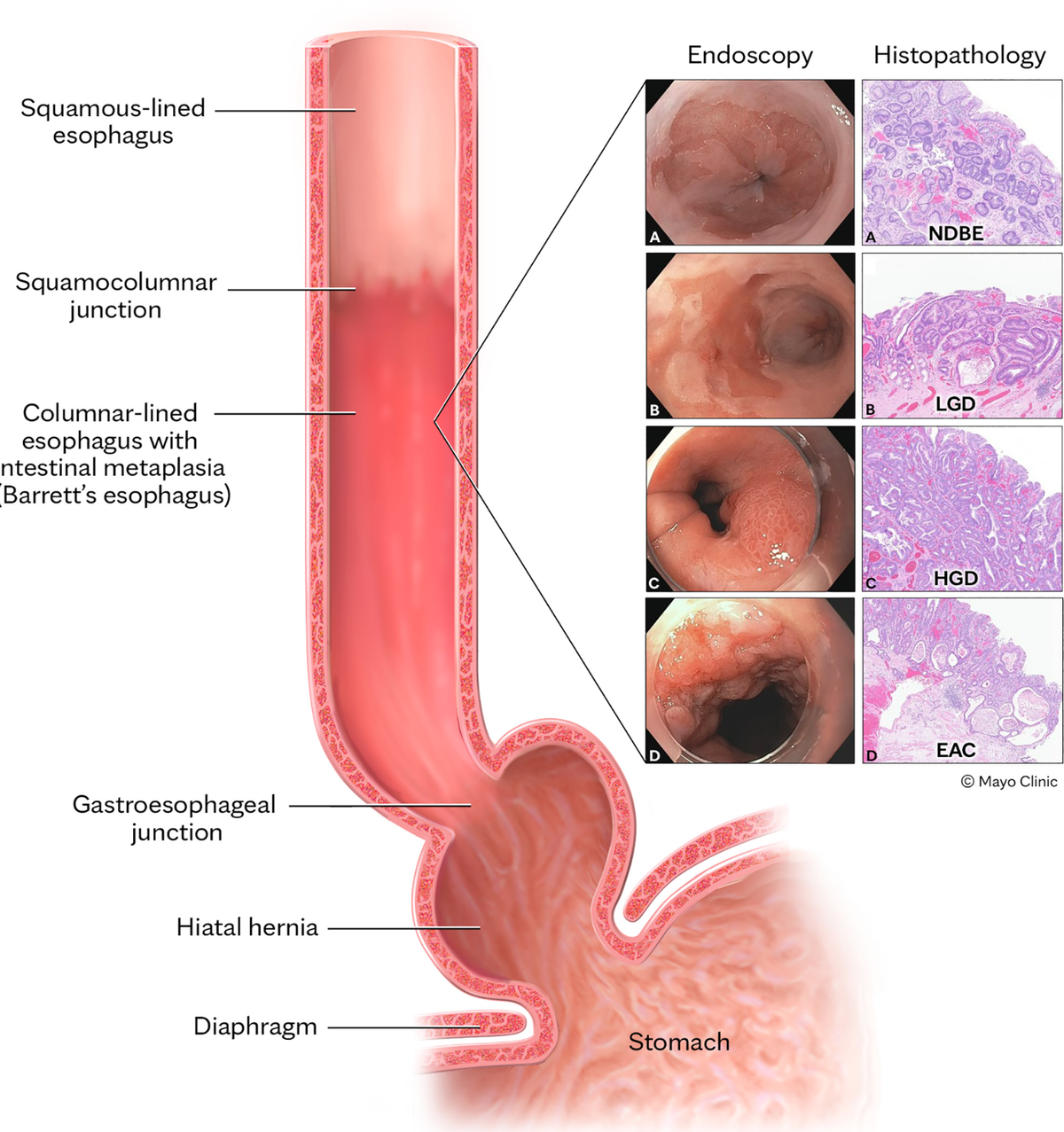
Esophageal adenocarcinoma (EAC), the primary form of esophageal cancer in the United States, is a lethal cancer with exponentially increasing incidence. Screening for Barrett esophagus (BE), the only known precursor to EAC, followed by endoscopic surveillance to detect dysplasia and early-stage EAC and subsequent endoscopic treatment (to prevent progression of dysplasia to EAC and to treat early-stage EAC effectively) is recommended by several society guidelines. Sedated endoscopy (the primary current tool for BE screening) is both invasive and expensive, limiting its widespread use. In this review, we aim to provide a comprehensive review of recent innovations in the nonendoscopic detection of BE and EAC. These include swallowable cell sampling devices combined with protein and epigenetic biomarkers (which are now guideline endorsed as alternatives to sedated endoscopy), tethered capsule endomicroscopy, emerging peripheral blood-sampled molecular biomarkers, and exhaled volatile organic compounds. We also summarize progress and challenges in assessing BE and EAC risk, which is an important complementary component of the process for the clinical implementation of these innovative nonendoscopic tools, and propose a new paradigm for the strategy to reduce EAC incidence and mortality.
Copyright © 2023 Mayo Foundation for Medical Education and Research. Published by Elsevier Inc. All rights reserved.